A coordination compound extending, through repeating coordination entities, in one dimension, but with cross-links between two or more individual chains, loops, or spiro-links, or a coordination compound extending through repeating coordination entities in two or three dimensions.
Note: The preferred and most widely understood term will likely be: "coordination polymer". However, IUPAC endorses also the use of the term "coordination network", even though it should be clear that these two terms are not synonymous and that coordination network is in fact a subset of coordination polymer.
Examples: Nets can be formed by cross-links between single chains as shown in Fig. 1, loops as shown in Fig. 2, or spiro-links, an example of which is displayed in Fig. 3, these type of compounds also belong to the coordination networks, while still extending only in one dimension and therefore also being 1D-coordination polymers. In Fig. 4, a classical 3D-coordination polymer is shown.
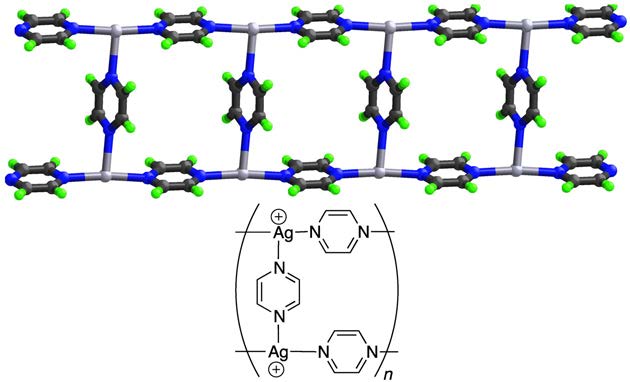
Figure 1: Example of cross-links forming a coordination network.
The weakly coordinating trifluoromethane-sulfonate anions are not shown.
Light grey: $\ce{Ag}$; blue: $\ce{N}$; grey: $\ce{C}$; green: $\ce{H}$.
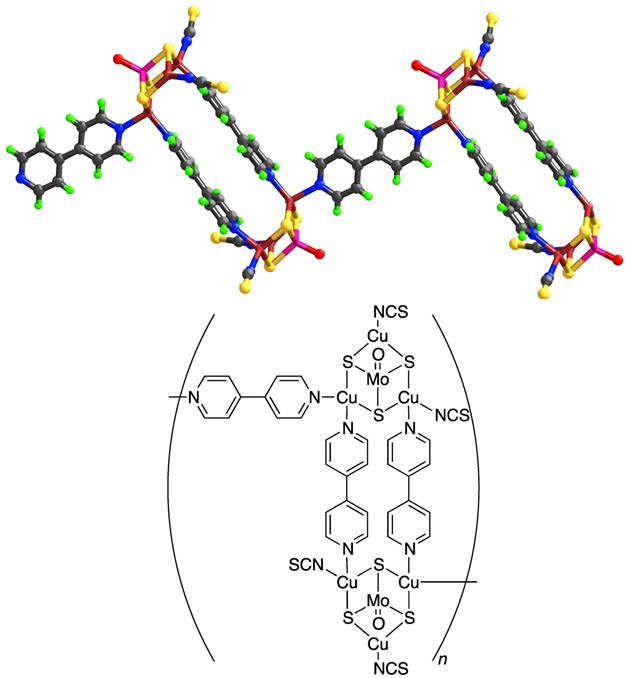
Figure 2: Example of loops forming a coordination network.
The ammonium cations and the aniline lattice molecules are not shown.
Mauve: $\ce{Mo}$; brown: $\ce{Cu}$; blue: $\ce{N}$, grey: $\ce{C}$; red: $\ce{O}$; green: $\ce{H}$.
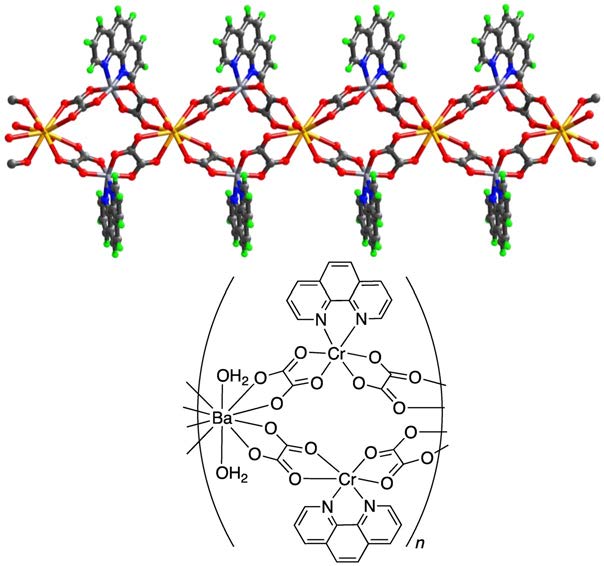
Figure 3: Example of spiro connections forming a coordination network.
The lattice water molecules are not shown.
Light grey: $\ce{Cr}$; yellow: $\ce{Ba}$; blue: $\ce{N}$; grey: $\ce{C}$; red: $\ce{O}$; green: $\ce{H}$.
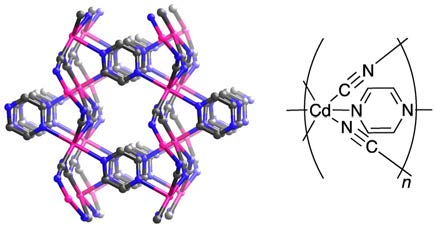
Figure 4: Example of a coordination network that is also a 3D-coordination polymer.
Hydrogen atoms have been omitted for clarity. Mauve: $\ce{Cd}$; blue: $\ce{N}$; grey: $\ce{C}$.
Source: PAC, 2013,
85, 1715. (
Terminology of metal–organic frameworks and coordination polymers (IUPAC Recommendations 2013)) on page 1717 [
Terms] [
Paper]



