https://doi.org/10.1351/goldbook.C00806
The electrically neutral species $\ce{H2C{:}}$ (methylene) and its derivatives, in which the carbon is covalently bonded to two univalent groups of any kind or a divalent group and bears two nonbonding electrons, which may be spin-paired (singlet state) or spin-non-paired (triplet state). In systematic name formation, carbene is the name of the parent hydride $\ce{C{:}H2}$ hence, the name dichlorocarbene for $\ce{C{:}Cl2}$. However, names for acyclic and cyclic hydrocarbons containing one or more divalent carbon atoms are derived from the name of the corresponding all-\(\lambda ^{4}\)-hydrocarbon using the suffix -ylidene. E.g. prop-2-en-1-ylidene, $\ce{H2C=CHCH{:}}$ ethenylidene, $\ce{H2C=C{:}}$; cyclohexylidene,
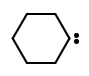
Subclasses of carbenes include acyl carbenes , imidoyl carbenes,
, imidoyl carbenes,  and vinyl carbenes.
and vinyl carbenes.
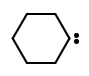
Subclasses of carbenes include acyl carbenes

